Does the Dose Still “Make the Poison” under Daubert?
Toxic tort litigation typically turns on a deceptively simple question: did a particular substance cause a particular harm? More fundamentally, is a particular substance harmful in the first place? In the legal context, courts traditionally have held to the old adage that “the dose makes the poison.” Under that view, no substance is per se harmful. Any substance can be harmful, but only after a certain threshold dose has been administered, and that dose can vary widely from substance to substance. For example, apple seeds contain cyanide compounds, but they are harmless unless ingested in massive quantities. Even water can be fatal if you drink enough.
Recent scientific evidence challenges this view, however, and indicates that some substances may be harmful at any level, and some substances’ harmful properties may not easily be correlated with the dose level. As courts consider this evidence, they may be swayed to accept evidence that would have otherwise been rejected under the “dose makes the poison” model.
“Dose-response curves” show the relationship between the dose of a substance and the harm it may cause. There are two general categories of dose-response curves:
- Monotonic.
- Non-monotonic.
Monotonic curves continually slope upward: increasing doses are increasingly harmful. Substances following this model are generally considered either “threshold” or “linear.” The threshold curve represents the “dose makes the poison” concept—a substance only becomes harmful after a certain threshold dose has been administered. For substances following a linear curve, any dose is harmful.
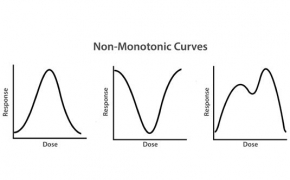
By contrast, a non-monotonic curve indicates that increasing levels of the dose are not necessarily related to increasing harm. What’s more, the level of response may not easily be correlated with the amount of the dose. Although not as clearly defined, there are several potential forms for nonmonotonic curves, as illustrated to the right.
If courts are willing to accept expert evidence that certain substances operate based on a non-threshold model—that is to say, they are either linear or non-monotonic—they may face a new generation of toxic tort suits based on expert testimony that goes beyond the traditional “dose makes the poison” framework.
Dose-Response and Daubert
In federal court and most state courts, expert testimony is evaluated under the Daubert standard. Daubert v. Merrell Dow Pharmaceuticals, 509 U.S. 579 (1993). Daubert requires trial judges to act as gatekeepers, ensuring that scientific expert testimony is relevant and reliable. Under Daubert, expert conclusions qualify as admissible scientific knowledge if they are derived from the scientific method. If the conclusions are not relevant or reliable or do not proceed from the scientific method, the testimony is inadmissible. Daubert has been codified as follows in Federal Rule of Evidence 702:
A witness who is qualified as an expert by knowledge, skill, experience, training, or education may testify in the form of an opinion or otherwise if:
- The expert’s scientific, technical, or other specialized knowledge will help the trier of fact to understand the evidence or to determine a fact in issue.
- The testimony is based on sufficient facts or data.
- The testimony is the product of reliable principles and methods.
- The expert has reliably applied the principles and methods to the facts of the case.
No Daubert court, to our knowledge, has examined expert testimony applying a non-monotonic, non-threshold dose-response curve. However, many Daubert courts have examined linear monotonic curves—meaning, the harm increases with increasing doses, but the dose need not reach a threshold: harm may occur at the lowest possible dosage level. In general, courts have relied on Daubert to reject expert testimony that these “low-dose” exposures can be harmful, on the grounds that such testimony is scientifically unreliable. For example, in Milward v. Acuity Specialty Prods. Group, 969 F. Supp. 2d 101 (D. Mass. 2013), the court excluded as “inadmissibly unreliable” the plaintiff’s claims that there is no safe level of benzene exposure.
Several courts have gone so far as to reject non-threshold models entirely. The U.S. Bankruptcy Court for the District of Delaware noted that the “the ‘no-threshold model . . . flies in the face of the toxicological law of dose-response . . . doesn’t satisfy Daubert, and doesn’t stand up to scientific scrutiny.” In re W.R. Grace & Co. 355 B.R. 462, 476 (Bankr. D. Del. 2006). The U.S. District for the District of Kansas was equally harsh, finding that the linear model “fails all of the Daubert reliability factors.” The model “cannot be falsified, nor can it be validated. To the extent that it has been subject to peer review and publication, it has been rejected by the overwhelming majority of the scientific community. It has no known or potential rate of error. It is merely a hypothesis . . . . In sum, it has no capacity to be of assistance to a jury in resolving the ultimate issues of a case.” Whiting v. Boston Edison Co., 891 F. Supp. 12, 25 (D. Mass. 1995).
On the other hand, not all courts have completely rejected non-threshold models. This is particularly true in the regulatory context, where courts are deferential to agency action. For example, a California state court upheld the California Air Resources Board’s use of a no-threshold model to establish the safe level of asbestos exposure as zero. Coalition for Reasonable Regulation of Naturally Occurring Substances v. Cal. Air Resources Bd., 19 Cal. Rptr. 3d 635, 641 (Cal. Ct. App. 2004). Courts can also focus on the causation side of the equation to avoid determining the validity of an expert’s proffered dose-response curve. The U.S. District for the Western District of Texas followed that path, declining to reject a proffered no-threshold model for uranium exposure out of hand, but instead tossed the plaintiffs’ scientific evidence because they had failed to show a causal connection between exposure and cancer. Cano v. Everest Minerals Corp., 362 F. Supp. 2d 814, 853–54 (W.D. Tex. 2005).
Recent Scientific Developments
Despite the judiciary’s general rejection of any models outside of the “dose makes the poison” framework, there is scientific evidence indicating that several categories of illness may be linked to non-threshold dose-response curves.
First, some genetic mutations can cause cancer at very low doses, following a linear dose-response curve. As described by the Reference Manual on Scientific Evidence, “In theory, the cancer-causing mutation to the genetic material of the cell can be produced by any one molecule of certain chemicals.” There is a risk—albeit a small one—that a single molecule would trigger the mutational process and cause cancer. For that reason, U.S. regulatory agencies have not eschewed linear models when evaluating cancer risks. As a measure of conservatism, U.S. government agencies presume that all carcinogens follow a linear response curve unless proven otherwise.
Of course, the regulatory context is very different from litigation: The agencies are taking precautionary measures to avoid general harm, while toxic tort litigation represents a specific effort to demonstrate that harm was caused in a specific person or group of people. For these reasons, the regulatory approach generally has been greeted with skepticism in the toxic tort context. The U.S. District Court for the District of Kansas noted that while the linear model may be appropriate for precautionary regulatory decision making, it is not acceptable in lawsuits that “must be resolved by reasonable conclusions based on the evidence, not by educated guesses.” Johnston v. United States, 597 F. Supp. 374 (D. Kan. 1984). In addition, not all cancers follow a mutational process. There is increasing evidence that some cancer-causing agents act through a phased, non-mutational process, and would, therefore, be better analyzed under the threshold dose-response model.
This scientific theory has been tested in several cases and generally has not survived. In Burleson v. Texas Department of Criminal Justice, 393 F.3d 577, 582 (5th Cir. 2004), a toxicologist testified that the primary risk factor for cancer is “the local microscopic dose of radiation that is received by the one cell that transforms into cancer.” Based on that scientific theory, the toxicologist opined that a prison inmate’s throat and lung cancer was most likely caused by inhalation of hazardous fumes in the prison welding workshop—and not by any other factor, including the inmate’s long history of smoking. The lower court found that the testimony failed under Daubert because:
- The theory had never been tested or submitted for peer review.
- The potential rate of error was high.
- Tt was not generally accepted in the scientific community.
- It was not relevant because there was no direct evidence of the level of the plaintiff’s exposure.
393 F.3d at 584. The Fifth Circuit affirmed, noting that, “scientific knowledge of the harmful level of exposure to a chemical, and knowledge that the plaintiff was exposed to such quantities are minimal facts necessary to sustain the plaintiffs’ burden.” Id. at 586.
Most recently, however, a California appeals court admitted expert testimony that “every exposure” to asbestos increases the risk of mesothelioma. Davis v. Honeywell International Inc., 245 Cal. App. 4th 477, 487 (Cal. App. 2016), petition for review pending. The court noted that mesothelioma can be both dose-dependent (i.e., risk increases as exposure increases) and every exposure can be a substantial factor in causing the disease: “The fact that mesothelioma is dose-dependent does not render Dr. Strauchen’s opinion that every exposure can be a substantial factor in causing the disease illogical.” Because the issue was a matter of “legitimate scientific debate,” the court would leave it up to the jury to weigh the evidence.
Second, endocrine disrupters—which can interfere with the human hormone system’s regulation of fertility and neural development—also have been shown to follow non-monotonic dose-response curves. EPA has found that some endocrine-disrupting chemicals may produce stronger toxic responses at low doses than at high doses. See EPA, "Endocrine Disruption Research: Testing for Potential Low-Dose Effects." Based on this evidence, some scientists have argued that it is improper to assume that harmful effects cannot occur at low-dose levels of endocrine exposure simply because no effects are seen at much higher levels. See Laura N. Vandenberg, et al., "Hormones and Endocrine-Disrupting Chemicals: Low-Dose Effects and Nonmonotonic Dose Responses," 33 Endocrine Reviews 378– 455 (June 2012).
In response to this body of research, EPA has investigated non-monotonic dose responses and endocrine disrupters. Most recently, EPA convened a working group to review the state of the science and determine the degree to which non-monotonic dose-response curves are evidenced in the scientific literature. The group prepared a draft paper, “State of the Science Evaluation: Nonmonotonic Dose Responses as They Apply to Estrogen Androgen, and Thyroid Pathways and EPA Testing and Assessment Procedures,” which was submitted to the National Academy of Sciences (NAS) on June 18, 2013. The NAS provided their findings and recommendations to EPA in May 2014, but EPA has not yet moved forward to finalize the paper.
The working group’s draft paper found that non-monotonic dose-response curves can occur in estrogen, androgen, and thyroid systems in ecological and mammalian populations. These effects are more commonly identified in vitro than in vivo, meaning that fetuses are most vulnerable to experiencing harmful effects at low doses. However, while these non-monotonic dose responses “do occur in biological systems,” they are “generally not common.” In addition, the group found that there is currently no reproducible evidence that non-monotonic dose responses occurring at low doses are predictive of adverse outcomes in humans. For these reasons, the working group found that EPA’s existing chemical testing and risk assessment procedures—which generally assume linear dose-response relationships—are still appropriate.
Daubert Compared with Frye
A minority of state courts have not adopted the Daubert standard, and instead apply the older Frye standard, which holds that scientific evidence is admissible only if the methodology upon which the expert’s opinion is based is “sufficiently established to have gained general acceptance in the particular field in which it belongs.” Frye v. United States, 293 F. 1013, 1014 (D.C. Cir. 1923).
There is some indication that Frye courts would be more willing to accept expert evidence of non-threshold dose curves. For example, although the Honeywell court did not explicitly cite Frye, this line of reasoning is in line with typical Frye analysis, and California courts have long declined to adopt Daubert (with the notable exception of a 2012 California Supreme Court case, Sargon Enterprises, Inc. v. University of Southern California, 55 Cal. 4th 747 (2012), in which the court appeared to adopt a standard similar to Daubert).
On the other hand, Frye requires courts to look to the general acceptance in the scientific community, and the science surrounding non-monotonic dose-response curves is still evolving. For that reason, some Frye courts might actually be more willing to reject non-monotonic evidence on the grounds that the evidence is not yet well-accepted in the scientific community. By contrast, a Daubert court would put greater weight on whether the evidence had been peer-reviewed and was scientifically reliable—by that standard, a single study could theoretically pass muster.
At this point, the scientific evidence of non-threshold dose-response curves is generally not adequate to survive true Daubert scrutiny. However, as scientific techniques evolve and the available evidence grows, non-threshold models may be increasingly reliable, and ultimately may be able to pass muster. In particular, given the large volume of ongoing asbestos litigation, and the favorable decision in the Honeywell case, plaintiffs’ attorneys in asbestos cases will likely continue to press linear dose-response models in their expert cases.
As for non-monotonic curves, the science surrounding endocrine disrupters is far less mature than asbestos research. However, this evolving area will likely be a source of intense litigation interest in the future. It is only a matter of time before such evidence comes before a court, which will have to reckon with the unusual nature of these substances. If courts admit expert testimony that minimal exposure to endocrine disrupters can cause serious harm to fetuses, there likely will be a deluge of litigation from the plaintiffs bar pressing claims against a wide variety of manufacturers and industry groups. Ultimately, both plaintiffs and defendants must track the scientific developments in this area carefully, and be sure to develop strong and thorough expert cases to defend their perspectives.
©2017. Published in Natural Resources & Environment, Vol. 31, No. 4, Spring 2017, by the American Bar Association. Reproduced with permission. All rights reserved.